Univ.-Prof. Dr. Alexander Loy

PUBLICATIONS AND CV
See here for a complete list of peer-reviewed papers, book chapters, and other publications.
A link to my CV is available at the bottom of this page.
Sulfur microorganisms
It is mainly through microbial activities that the sulfur cycle is intimately intertwined with the cycling of other elements such as carbon or nitrogen. My group is interested in the evolution of sulfur microorganisms, their yet largely unexplored diversity, distribution, and ecological function in various environments, and their roles as symbionts of animals and humans.
Intestinal microbiota
 The symbiotic intestinal microbiota provides important benefits to its animal and human host such as breakdown of complex nutritional substrates and protection against pathogens. Changes in the intestinal microbiota are, however, associated with many diseases. We are interested in the ecophysiology of the gut microbiota in health and disease. In the context of various diseases such as inflammatory bowel disease, cancer or intestinal infections, we specifically aim to answer how nutrition impacts the gut microbiota and which/how individual community members influence physiology and health of their host.
The symbiotic intestinal microbiota provides important benefits to its animal and human host such as breakdown of complex nutritional substrates and protection against pathogens. Changes in the intestinal microbiota are, however, associated with many diseases. We are interested in the ecophysiology of the gut microbiota in health and disease. In the context of various diseases such as inflammatory bowel disease, cancer or intestinal infections, we specifically aim to answer how nutrition impacts the gut microbiota and which/how individual community members influence physiology and health of their host.
Individual cells of the intestinal microbiota either compete for substrates due to functional overlap or cooperate in syntrophic substrate degradation, resulting in a dynamic network of synergistic and antagonistic physiological interactions that are reciprocally influenced by the fluctuating physical and chemical environment of the gut. Here, in particular single cell isotope labeling tools promise yet unavailable detailed insights into host- or diet-derived substrate partitioning and niche competition among individual intestinal microbiota members, including incoming pathogens (Loy and Berry, 2018. Trends Microbiol, Stecher, Berry, and Loy. 2013. FEMS Microbiol Rev).
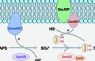
One focus of our work is the evolution and function of sulfur compounds-utilizing and hydrogen sulfide-producing gut microorganisms because a balanced sulfur metabolism is crucial for intestinal homeostasis and host health. Furthermore, some pathogens such as Salmonella influence the intestinal sulfur metabolism to selectively support their outgrowth and fitness, but the role of the indigenous sulfur microbiota during these intestinal infections is unknown.
Techniques
To accomplish our research goals, we use a holistic approach that combines molecular biology, genomic, and isotope-labeling techniques for analyses of microbial isolates and communities (ranging from defined assemblages of known strains to naturally complex microbiotas in in situ-like microcosms and the environment)
Selected methods: highly parallel amplicon next-generation-sequencing of functional and phylogenetic marker genes, genomics and postgenomic analyses (meta-omics), DNA microarrays, fluorescence in situ hybridization, real-time PCR, Raman microspectroscopy, NanoSIMS, and phylogenetics/-genomics.
Together with other DOME members, we are continuously developing novel molecular and isotope-labeling methods for structure-function analysis of complex microbial communities.
Research Topics
- Evolution and ecology of sulfur microorganisms, with focus on sulfate-reducing microorganisms
- Function of the intestinal microbiota in health and disease
- Development of molecular and isotope-labeling methods for studying uncultivated microorganisms in their natural environment
CURRENT PROJECTS
- The wetland sulfur microbiome
- The MetaBac research platform - secondary metabolomes of bacterial communities
- Establishing defined communities of Antarctic soil bacteria as potential sources of antimicrobials
FINISHED PROJECTS
- Physiological interactions of Salmonella and the intestinal microbiota
- Interspecies hydrogen transfer in the mammalian gut
- Psychrophilic sulfate-reducing microorganisms in Arctic marine sediments
- Missing links in the marine sulfur cycle - microorganisms that utilize organic sulfur compounds and anorganic sulfur compounds of intermediate oxidation states
- Nutrition and the intestinal microbiota-host symbiosis
PREVIOUS SELECTED projects
Intestinal microbiota - Inflammation and a new method for analysis of microbial physiology in vivo
 Inflammatory bowel diseases are characterized by an altered gut microbiota composition. By using mouse colitis models, we identified indicator phylotypes for health state (Berry et al. 2012. ISME J, Berry et al. 2015. Front Microbiol) and revealed characteristic changes in the microbial transcriptome and interactions with the host (Schwab et al. 2014. ISME J). In a clinical pilot study, we showed that treatment success and the temporal fecal microbiota dynamics varied among ulcerative colitis patients after receiving fecal microbiota transplantation from healthy donors as an intervention therapy (Angelberger et al. 2013. Am J Gastroenterol). By developing a novel single-cell, isotope-probing method, we could identify, for the first time, important foragers of mouse host-derived protein compounds in vivo and show that the host-foraging phenotype of these bacteria is quantitatively dependent on the complexity of the intestinal microbiota (Berry et al. 2013. PNAS, Stecher et al. 2013. FEMS Microbiol Rev).
Inflammatory bowel diseases are characterized by an altered gut microbiota composition. By using mouse colitis models, we identified indicator phylotypes for health state (Berry et al. 2012. ISME J, Berry et al. 2015. Front Microbiol) and revealed characteristic changes in the microbial transcriptome and interactions with the host (Schwab et al. 2014. ISME J). In a clinical pilot study, we showed that treatment success and the temporal fecal microbiota dynamics varied among ulcerative colitis patients after receiving fecal microbiota transplantation from healthy donors as an intervention therapy (Angelberger et al. 2013. Am J Gastroenterol). By developing a novel single-cell, isotope-probing method, we could identify, for the first time, important foragers of mouse host-derived protein compounds in vivo and show that the host-foraging phenotype of these bacteria is quantitatively dependent on the complexity of the intestinal microbiota (Berry et al. 2013. PNAS, Stecher et al. 2013. FEMS Microbiol Rev).
Selected publications on this theme:
- Berry D, Kuzyk O, Rauch I, Heider S, Schwab C, Hainzl E, Decker T, Müller M, Strobl B, Schleper C, Urich T, Wagner M, Kenner L, Loy A. 2015. Intestinal microbiota signatures associated with inflammation history in mice experiencing recurring colitis. Front Microbiol. 6: 1408.
- Schwab C, Berry D, Rauch I, Rennisch I, Ramesmayer J, Hainzl E, Heider S, Decker T, Kenner L, Müller M, Strobl B, Wagner M, Schleper C, Loy A, Urich T. 2014. Longitudinal study of murine microbiota activity and interactions with the host during acute inflammation and recovery. ISME J. 8: 1101-1114.
- Berry D, Stecher B, Schintlmeister A, Reichert J, Brugiroux S, Wildd B, Wanek W, Richter A, Rauch I, Decker T, Loy A, Wagner M. 2013. Host-compound foraging by intestinal microbiota revealed by single-cell stable isotope probing. Proc. Natl. Acad. Sci. USA 110: 4720-4725.
- Stecher B, Berry D, Loy A. 2013. Colonization resistance and microbial ecophysiology: Using gnotobiotic mouse models and single-cell technology to explore the intestinal jungle. FEMS Microbiol. Rev. 37: 793-829.
- Angelberger S, Reinisch W, Makristathis A, Lichtenberger C, Dejaco C, Papay P, Novacek G, Trauner M, Loy A, Berry D. 2013. Temporal bacterial community dynamics vary among ulcerative colitis patients after fecal microbiota transplantation. Am. J. Gastroenterol. 108: 1620-1630.
- Berry D, Schwab C, Milinovich G, Reichert J, Ben Mahfoudh K, Decker T, Engel M, Hai B, Hainzl E, Heider S, Kenner L, Müller M, Rauch I, Strobl B, Wagner M, Schleper C, Urich T, Loy A. 2012. Phylotype-level 16S rRNA analysis reveals new bacterial indicators of health state in acute murine colitis. ISME J. 6: 2091-106.
Sulfate-reducing microorganisms mitigate methane emissions from wetlands
 Wetlands are important carbon sinks but at the same time major global sources of the greenhouse gas methane. A hidden sulfur cycle in wetlands allows sulfite/sulfate-reducing microorganism to successfully compete with methanogenic archaea and thereby to reduce methane emissions (Pester et al. 2012. Front Microbiol). While novel, uncultivated lineages dominate dsrAB diversity in wetlands (Steger et al. 2011. Appl Environ Microbiol), we identified rare microbiota members (including Desulfosporosinus) as important sulfate reducers in an acidic peatland by comparative 16S rRNA gene- and dsrAB-stable isotope probing (Pester et al. 2010. ISME J). To further our understanding of sulfate reducer ecophysiology and genomics in peatlands, we are investigating the species interactions (Hausmann et al. 2016. ISME J) and metatranscriptome dynamics in microcosms under in situ-like conditions (Hausmann et al. 2018. ISME J, Hausmann et al. 2019. mBio).
Wetlands are important carbon sinks but at the same time major global sources of the greenhouse gas methane. A hidden sulfur cycle in wetlands allows sulfite/sulfate-reducing microorganism to successfully compete with methanogenic archaea and thereby to reduce methane emissions (Pester et al. 2012. Front Microbiol). While novel, uncultivated lineages dominate dsrAB diversity in wetlands (Steger et al. 2011. Appl Environ Microbiol), we identified rare microbiota members (including Desulfosporosinus) as important sulfate reducers in an acidic peatland by comparative 16S rRNA gene- and dsrAB-stable isotope probing (Pester et al. 2010. ISME J). To further our understanding of sulfate reducer ecophysiology and genomics in peatlands, we are investigating the species interactions (Hausmann et al. 2016. ISME J) and metatranscriptome dynamics in microcosms under in situ-like conditions (Hausmann et al. 2018. ISME J, Hausmann et al. 2019. mBio).
Selected publications on this theme:
- Hausmann B, Pelikan C, Herbold CW, Köstlbacher S, Albertsen M, Eichorst SA, Glavina del Rio T, Huemer M, Nielsen PH, Rattei T, Stingl U, Tringe SG, Trojan D, Wentrup C, Woebken D, Pester M, and Loy A [2018] Peatland Acidobacteria with a dissimilatory sulfur metabolism. ISME J. doi:10.1038/s41396-018-0077-1
- Hausmann B, Knorr K-H, Schreck K, Tringe SG, del Rio TG, Loy A, and Pester M [2016] Consortia of low-abundance bacteria drive sulfate reduction-dependent degradation of fermentation products in peat soil microcosms. ISME J. doi: 10.1038/ismej.2016.42
- Pester M, Knorr K-H, Friedrich MW, Wagner M, Loy A. 2012. Sulfate-reducing microorganisms in wetlands - fameless actors in carbon cycling and climate change. Frontiers Microbiol. 3: 72.
- Steger D, Wentrup C, Braunegger C, Deevong P, Hofer M, Richter A, Baranyi C, Pester M, Wagner M, Loy A. 2011. Microorganisms with novel dissimilatory (bi)sulfite reductase genes are widespread and part of the core microbiota in low-sulfate peatlands. Appl. Environ. Microbiol. 77: 1231-1242.
- Pester M, Bittner N, Deevong P, Wagner M, Loy A. 2010. A 'rare biosphere' microorganism contributes to sulfate reduction in a peatland. ISME J. 12: 1591-1602.
Evolution & environmental diversity of an ancient enzyme and discovery of novel sulfur microorganisms
 The evolution of DsrAB-type dissimilatory sulfite reductase has been influenced by lateral gene transfer but surprisingly few gene duplication events. This enzyme and its genes have been successfully applied, within certain limits, as diagnostic markers for environmental studies of sulfite/sulfate-reducing and some sulfur-oxidizing microorganisms (Sabehi et al. 2005. PLoS Biol, Loy et al. 2009. Environ Microbiol, Gruber-Vodicka et al. 2011. PNAS). Amplicon sequencing and genome-centric metagenomics of diverse environments has shown that DsrAB-based dissimilatory sulfur metabolism is more widespread among extant bacterial and archaeal phyla than previously recognized (Müller et al. 2015. ISME J, Anantharaman et al. 2018. ISME J). Our publically available dsrAB/DsrAB database, a new multi-level classification system, and new primers also allow for large-scale dsrAB ecology studies using multiplex amplicon sequencing (Pelikan et al. 2016. Environ Microbiol).
The evolution of DsrAB-type dissimilatory sulfite reductase has been influenced by lateral gene transfer but surprisingly few gene duplication events. This enzyme and its genes have been successfully applied, within certain limits, as diagnostic markers for environmental studies of sulfite/sulfate-reducing and some sulfur-oxidizing microorganisms (Sabehi et al. 2005. PLoS Biol, Loy et al. 2009. Environ Microbiol, Gruber-Vodicka et al. 2011. PNAS). Amplicon sequencing and genome-centric metagenomics of diverse environments has shown that DsrAB-based dissimilatory sulfur metabolism is more widespread among extant bacterial and archaeal phyla than previously recognized (Müller et al. 2015. ISME J, Anantharaman et al. 2018. ISME J). Our publically available dsrAB/DsrAB database, a new multi-level classification system, and new primers also allow for large-scale dsrAB ecology studies using multiplex amplicon sequencing (Pelikan et al. 2016. Environ Microbiol).
Notably, abundant ocean viruses also carry microbial sulfur genes and potentially manipulate the sulfur metabolism of their microbial hosts (Roux et al. 2016. Nature).
Selected publications on this theme:
- Anantharaman K, Hausmann B, Jungbluth SP, Kantor RS, Lavy A, Warren LA, Rappé MS, Pester M, Loy A, Thomas BC, and Banfield JF [2018] Expanded diversity of microbial groups that shape the dissimilatory sulfur cycle. ISME J. doi:10.1038/s41396-018-0078-0
- Roux S, Brum JR, Dutilh BE, Sunagawa S, Duhaime MB, Loy A, Poulos BT, Solonenko N, Lara E, Poulain J, Pesant S, Kandels-Lewis S, Dimier C, Picheral M, Searson S, Cruaud C, Alberti A, Duarte CM, Gasol JM, Vaque D, Tara Oceans Coordinators, Bork P, Acinas SG, Wincker P, and Sullivan MB. 2016. Ecogenomics and potential biogeochemical impacts of globally abundant ocean viruses. Nature. 537: 689–693. doi: 10.1038/nature19366
- Pelikan C, Herbold CW, Hausmann B, Müller AL, Pester M, Loy A. 2016. Diversity analysis of sulfite- and sulfate-reducing microorganisms by multiplex dsrA and dsrB amplicon sequencing using new primers and mock community-optimized bioinformatics. Environ Microbiol. doi: 10.1111/1462-2920.13139
- Müller AL, Kjeldsen KU, Rattei T, Pester M, Loy A. 2015. Phylogenetic and environmental diversity of DsrAB-type dissimilatory (bi)sulfite reductases. ISME J. 9: 1152-1165.
- Gruber-Vodicka HR, Dirks U, Leisch N, Baranyi C, Stoecker K, Bulgheresi S, Heindl NR, Horn M, Lott C, Loy A, Wagner M, Ott J. 2011. Paracatenula, an ancient symbiosis between thiotrophic Alphaproteobacteria and catenulid flatworms. Proc. Natl. Acad. Sci. USA 108: 12078-12083.
- Loy A, Duller S, Baranyi C, Mußmann M, Ott J, Sharon I, Béjà O, Le Paslier D, Dahl C, Wagner M. 2009. Reverse dissimilatory sulfite reductase as phylogenetic marker for a subgroup of sulfur-oxidizing prokaryotes. Environ. Microbiol. 11: 289-299.
- Sabehi G#, Loy A#, Jung KH, Partha R, Spudich JL, Isaacson T, Hirschberg J, Wagner M, Beja O. 2005. New insights into metabolic properties of marine bacteria encoding proteorhodopsins. PLoS Biol. 3: e273. #equal contribution
Dormant microbes provide clues about passive dispersal of microorganisms in the ocean
 The combined effects of passive dispersal and environmental selection influence microbial biogeography, but the contribution of either factor is difficult to discern. Inactive spores of thermophilic bacteria that are present in the cold seabed are not subject to environmental selection and thus ideal dispersal indicators (Hubert et al. 2009. Science, de Rezende et al. 2013. ISME J). These spore-forming microorganisms likely originate from hot anoxic environments in the sea floor or the ocean crust such as hydrothermal vents and their overlying sediments, or gas and oil reservoirs. We showed that individual phylotypes of thermophilic spores have various patterns of spatial distribution in the world oceans and were dispersal limited to different degrees. The overall distribution pattern of thermophilic spores suggests that the impact of passive dispersal on marine microbial biogeography is controlled by the connectivity of local water masses to ocean circulation (Müller et al. 2014. ISME J, Hanson et al. 2019. Front Microbiol).
The combined effects of passive dispersal and environmental selection influence microbial biogeography, but the contribution of either factor is difficult to discern. Inactive spores of thermophilic bacteria that are present in the cold seabed are not subject to environmental selection and thus ideal dispersal indicators (Hubert et al. 2009. Science, de Rezende et al. 2013. ISME J). These spore-forming microorganisms likely originate from hot anoxic environments in the sea floor or the ocean crust such as hydrothermal vents and their overlying sediments, or gas and oil reservoirs. We showed that individual phylotypes of thermophilic spores have various patterns of spatial distribution in the world oceans and were dispersal limited to different degrees. The overall distribution pattern of thermophilic spores suggests that the impact of passive dispersal on marine microbial biogeography is controlled by the connectivity of local water masses to ocean circulation (Müller et al. 2014. ISME J, Hanson et al. 2019. Front Microbiol).
Selected publications on this theme:
- Müller AL, de Rezende JR, Hubert C, Kjeldsen KU, Lagkouvardos I, Berry D, Jørgensen BB, Loy A. 2014. Endospores of thermophilic bacteria as tracers of microbial dispersal by ocean currents. ISME J. 8: 1153-1165.
- de Rezende JR, Kjeldsen KU, Hubert C, Finster K, Loy A, Jørgensen BB. 2013. Dispersal of thermophilic Desulfotomaculum endospores to Baltic Sea sediments over thousands of years. ISME J. 7: 72-84.
- Hubert C, Loy A, Nickel M, Arnosti C, Baranyi C, Brüchert V, Ferdelman T, Finster K, Christensen FM, Rosa de Rezende J, Vandieken V, Jørgensen BB. 2009. A constant flux of diverse thermophilic bacteria into the cold Arctic seabed. Science 325: 1541-1544.
JOINING THE TEAM
Information on open research positions can be found here. If you are interested in joining our team with your own fellowship, please check out our PhD & postdoc program and get in touch with Alex for details.